Providing a pathway to potent pain therapy without the risk of addiction and serious side effects.
Phoenix is a pre-clinical pharmaceutical company driven by a mission to bring potent, safe pain therapies to market without risk of abuse and addiction.
THE PROBLEM
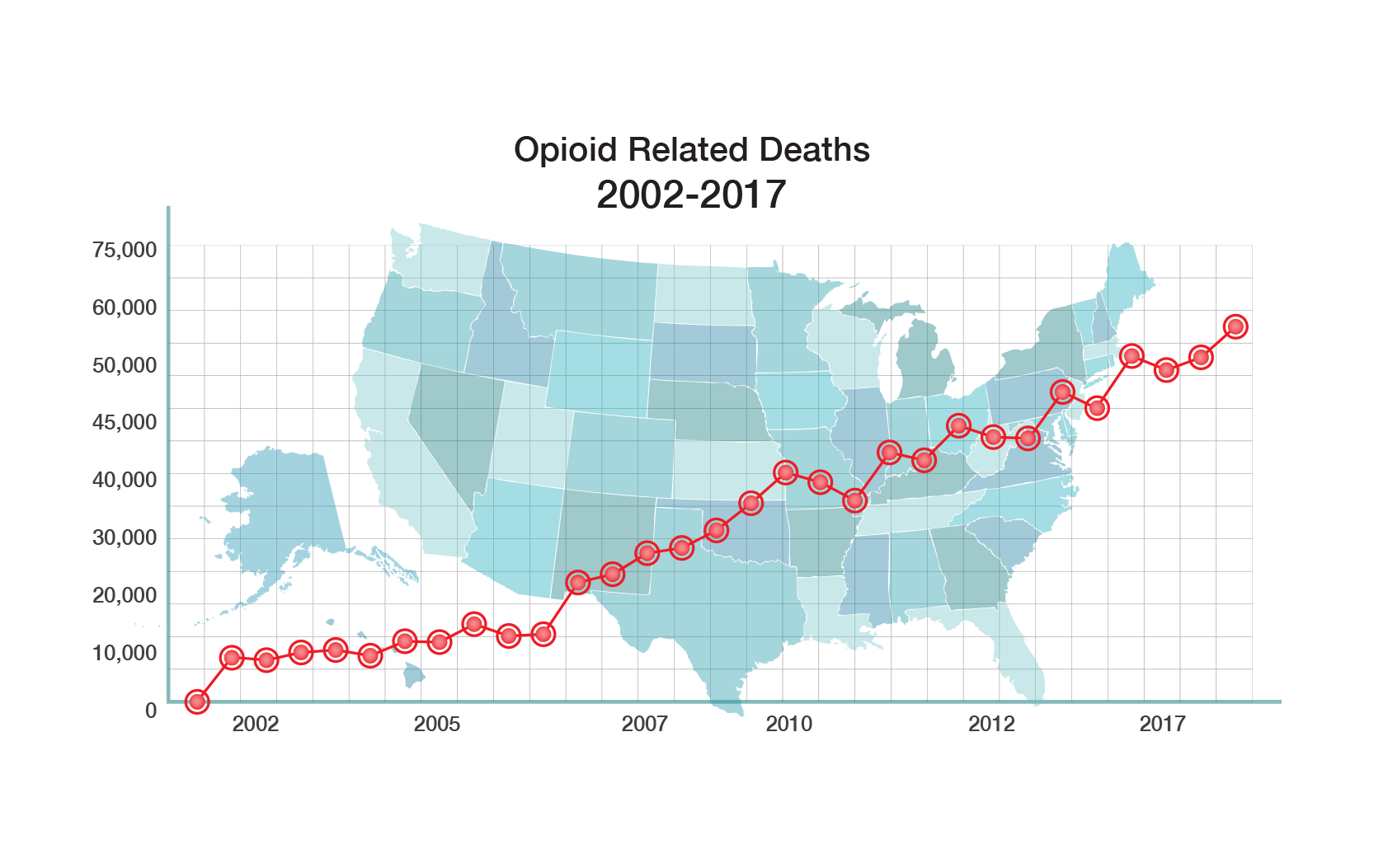
Opioids are the most widely prescribed drugs for treatment of moderate to severe pain. They are also the most powerful analgesics for treatment of acute and chronic pain. However, their use is plagued by serious side effects, including abuse and addiction, severe withdrawal, constipation, respiratory depression, and death from overdose.
Millions of Americans are seriously addicted to opioids – and it is a devastating addiction. There is an immense unmet medical need to comprehensively address the abuse, addiction and mortality associated with opiates.